|
Guillain-Barré Syndrome Following the BNT162b2 mRNA COVID-19 Vaccine
Authors:
Hussein A. Algahtani, 1 , Bader H. Shirah, 2 , Yazeed K. Albeladi, 3 , Reyadh K. Albeladi, 3 ,
1 Neurology Section, Department of Medicine, King Abdulaziz Medical City, Jeddah, Saudi Arabia; King Abdullah International Medical Research Center, Jeddah, Saudi Arabia; College of Medicine, King Saud bin Abdulaziz University for Health Sciences, Jeddah, Saudi Arabia
2 Department of Neuroscience, King Faisal Specialist Hospital & Research Centre, Jeddah, Saudi Arabia
3 College of Medicine, Jeddah University, Jeddah, Saudi Arabia
Corresponding Author:
Hussein Algahtani
keywords: Guillain-Barré syndrome; COVID-19; Vaccine; BNT162b2; mRNA
Abstract
Purpose: The onset of the COVID-19 (SARS-CoV-2) pandemic in December 2019 created the need for multiple scientific research activities and clinical trials in an attempt to find solutions to mitigate the impact of the virus. One of the important tools to combat the virus is the development of vaccination programs. All types of vaccines have been associated with a mild to severe risk of neurological adverse events. One of these severe adverse events is Guillain-Barré syndrome.
Case Report: Here, we describe a case of Guillain-Barré syndrome after the first dose of the BNT162b2 mRNA COVID-19 vaccine and review the literature to increase the current knowledge regarding this complication.
Conclusion: Guillain-Barré syndrome after COVID-19 vaccination is responsive to treatment. The benefits of administering the vaccine outweigh the risks. Due to the negative impact of COVID-19, it is essential to recognize the development of neurological complications that are potentially associated with vaccination, including Guillain-Barré syndrome.
Keywords: Guillain-Barré syndrome; COVID-19; Vaccine; BNT162b2; mRNA.
INTRODUCTION
The onset of the COVID-19 (SARS-CoV-2) pandemic in Wuhan, China in December 2019 created the need for conducting multiple scientific research activities and clinical trials in an attempt to find solutions to mitigate the catastrophic impact of the virus (1). One of the important tools to combat this highly infectious agent and its complications has been the development of efficacious vaccination programs (2). Several vaccines from different manufacturers have been developed to fight this deadly pandemic using different methodologies, including vector-based vaccines, COVID-19 mRNA-based vaccines, inactivated or attenuated virus vaccines, and protein-based vaccines (3). All types of vaccines are associated with a risk of neurological adverse events ranging from mild to severe (4). One of these important severe adverse events is Guillain-Barré syndrome (5. In this article, we describe a case of Guillain-Barré syndrome after the first dose of the BNT162b2 mRNA COVID-19 vaccine and review the literature.
CASE REPORT
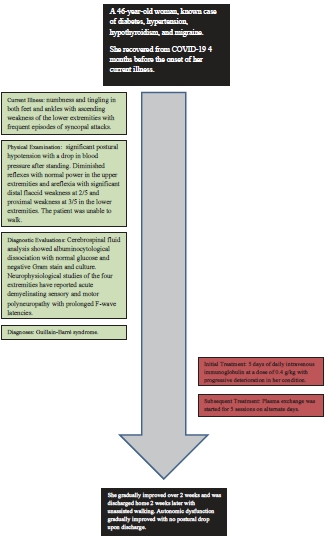
A 46-year-old woman presented to the emergency room complaining of numbness and tingling in both feet and ankles with ascending weakness of the lower extremities five days after receiving the first dose of the BNT162b2 mRNA COVID-19 vaccine. Immediately after vaccination, she was asymptomatic with no fever, myalgia, or flu-like symptoms. The weakness started initially in the left foot, progressed over 24h to the right foot, and then ascended rapidly to the degree that she was not able to stand without assistance three days later. The patient complained of frequent episodes of syncopal attacks while standing. There was no dysarthria, dysphagia, diplopia, facial weakness, sensory level, or urinary incontinence. She had diabetes, hypertension, hypothyroidism, and migraine. She recovered from COVID-19 four months before the onset of her current illness. Her social and family history was unremarkable.
Upon examination, she was conscious, oriented, and alert with a Glasgow coma scale score of 15/15. The blood pressure at presentation was 90/60 mmHg which normalized after fluid resuscitation. There was significant postural hypotension with a drop in blood pressure after standing, even after normalization of the blood pressure. There was no fever; however, sinus tachycardia was consistently present in the range of 100-120 beats per minute. Neurological examination showed normal cranial nerves II to XII, diminished deep tendon reflexes with normal power in the upper extremities, and areflexia with significant distal flaccid weakness at 2/5 and proximal weakness at 3/5 in the lower extremities in the Medical Research Council (MRC) scale. The patient was unable to walk due to weakness and postural hypotension. Sensory examination showed altered light touch, pinprick sensation, and vibration in a stocking distribution.
General hematological, biochemical, endocrine, infectious, tumor marker, and autoimmune disease screening results were unremarkable. Cerebrospinal fluid analysis showed one white blood cell with a high protein concentration of 1.39 g/L indicating albuminocytological dissociation with normal glucose and negative Gram stain and culture. Magnetic resonance imaging (MRI) of the brain and whole spine was unremarkable. Nerve conduction study of the four extremities reported acute demyelinating sensory and motor polyneuropathy with prolonged F-wave latencies.
Despite difficulties with blood pressure readings, the patient received five days of daily intravenous immunoglobulin at a dose of 0.4 g/kg with progressive deterioration in her condition including dysautonomia (resting sinus tachycardia and postural hypotension). Given the lack of response to intravenous immunoglobulin, plasma exchange was started one day later for five sessions on alternate days.
She gradually improved over two weeks and was discharged home two weeks later with unassisted walking. Autonomic dysfunction gradually improved with no postural drop upon discharge.
DISCUSSION
Several vaccines have been approved by different regulatory agencies worldwide based on randomized, blinded, controlled trials that started in December 2020. These approved vaccines include vector-based, COVID-19 mRNA-based, inactivated or attenuated virus, and protein-based vaccines. Vector-based vaccines include a recombinant chimpanzee adenovirus vector encoding the spike glycoprotein of SARS-CoV-2 (ChAdOx1 nCov-19) and a recombinant adenovirus type 26 vector encoding the SARS-CoV-2 spike protein (Ad26.COV2.S). Two COVID-19 mRNA-based vaccines that encode the spike protein antigen of SARS-CoV-2 encapsulated in lipid nanoparticles (BNT162b2 and mRNA-1273) have been approved. The other 2 vaccine types included inactivated or attenuated virus vaccines (PiCoVacc, BBIBP-CorV, BBV152) and protein-based vaccines (NVX-CoV2373). Upon reviewing the literature, most cases of Guillain- Barré syndrome following COVID-19 vaccination were related to vector-based vaccines (around two-thirds) and COVID-19 mRNA-based vaccines (one-third). Few cases have been reported for other types (3).
Guillain-Barré syndrome is a rapidly progressive immune-mediated polyradiculoneuropathy that classically presents with symmetric paresthesia, limb weakness, and areflexia (6). It is commonly triggered by infections with Campylobacter jejuni, Epstein-Barr virus, cytomegalovirus, influenza, and Mycoplasma pneumoniae. In addition, it can be triggered by vaccination, including hepatitis B, tetanus, polio, meningitis, rabies, and influenza (7). It is usually diagnosed by clinical evaluation assisted by neurophysiological testing and application of diagnostic criteria such as the Brighton criteria (8). There are several variants of this syndrome, and in up to 30% of the cases, respiratory weakness may progress to respiratory failure. It is critical to rapidly recognize the signs and symptoms of this disorder since most patients will require admission to expedite work-up, initiate immunomodulatory therapy, and monitor for complications including autonomic and respiratory compromise (9).
In our case, although a direct causal relationship between COVID-19 vaccines and Guillain-Barré syndrome could not be confirmed, the time interval between vaccination and onset of symptoms of Guillain-Barré syndrome was five days, which suggests a possible link with immunization. This time window is consistent with the previously reported duration of preceding triggering events leading to the development of an immune- mediated attack. In addition, the absence of any other triggering factors in our case confirms this relationship. The exact pathophysiological mechanism of Guillain- Barré syndrome following COVID-19 vaccination is poorly understood. The most plausible theory explaining this neurological complication is molecular mimicry, in which the body recognizes certain types of proteins, such as the SARS-CoV-2 spike protein, which is synthesized in the body or included in the vaccine. These proteins bind to sialic acid-containing glycoproteins and gangliosides on cell surfaces with subsequent cross-reactions and attack by the immune system (10).
The incidence of Guillain-Barré syndrome following COVID-19 vaccination has not been well estimated. This is because of the lack of population studies and limited literature, as most of the published research consists of case reports and series. Upon reviewing the literature, fewer than 300 cases have been reported with only three cohort studies (11-13). In our patient, the preceding COVID-19 vaccination with a temporal relationship may suggest a possible contribution of the vaccination to the development of Guillain-Barré syndrome. The risk of developing COVID-19 vaccine-associated Guillain-Barré syndrome remains very low given the huge number of people vaccinated worldwide. To fight against this deadly global pandemic, the public health systems in different countries decided that widespread vaccination is inevitable despite the scarcity of epidemiological data about the safety and complications of vaccination (14). This trust has proven to be logical and correct since the pandemic almost ended. As of late December 2021, approximately 280 million cases of COVID-19 have been reported, with approximately 5 million deaths (15).
CONCLUSION
We report a case of Guillain-Barré syndrome following the BNT162b2 mRNA COVID-19 vaccine to increase the current knowledge regarding this complication, which is responsive to treatment, and to elaborate on the benefits of administering the vaccine, which outweigh the risks. What makes our case unique is the severe autonomic symptoms, lack of response to intravenous immunoglobulin, and excellent outcome after plasma exchange. Most of the cases of Guillain-Barré syndrome related to vaccination were treated with intravenous immunoglobulin. The COVID-19 pandemic has negatively impacted the physical, social, and economic aspects of many societies. It is essential to recognize the development of neurological complications that are potentially associated with vaccination, including Guillain-Barré syndrome.
REFERENCES
- Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in china, 2019. N Engl J Med. 2020;382(8):727-733.
- Li YD, Chi WY, Su JH, Ferrall L, Hung CF, Wu Coronavirus vaccine development: from SARS and MERS to COVID-19. Journal of Biomedical Science. 2020;27(1):104.
- Garg RK, Paliwal Spectrum of neurological complications following COVID-19 vaccination. Neurol Sci. Published online October 31, 2021.
- Finsterer J. Neurological side effects of SARS-CoV-2 Acta Neurol Scand. 2022;145(1):5-9.
- Lahoz Fernandez PE, Miranda Pereira J, Fonseca Risso I, et Guillain-Barre syndrome following COVID-19 vaccines: A scoping review. Acta Neurol Scand. Published online December 29, 2021.
- Goodfellow JA, Willison Guillain-Barré syndrome: a century of progress. Nat Rev Neurol. 2016;12(12):723-731.
- Leonhard SE, Mandarakas MR, Gondim FAA, et Diagnosis and management of Guillain– Barré syndrome in ten steps. Nat Rev Neurol. 2019;15(11):671-683.
- Ghazanfar H, Qazi R, Ghazanfar A, Iftekhar Significance of brighton criteria in the early diagnosis and management of guillain-barré syndrome. Cureus. 12(5):e8318.
- Nguyen TP, Taylor RS. Guillain barre syndrome. In: StatPearls Publishing; 2022. Accessed February 2, 2022. http://www.ncbi.nlm.nih.gov/books/ NBK532254
- Koike H, Katsuno M. Emerging infectious diseases, vaccines and Guillain-Barré syndrome. Clin Exp Published online May 17, 2021.
- García-Grimshaw M, Michel-Chávez A, Vera- Zertuche JM, et Guillain-Barré syndrome is infrequent among recipients of the BNT162b2 mRNA COVID-19 vaccine. Clin Immunol. 2021;230:108818.
- Li X, Ostropolets A, Makadia R, et al. Characterizing the incidence of adverse events of special interest for COVID-19 vaccines across eight countries: a multinational network cohort medRxiv. Published online April 17, 2021:2021.03.25.21254315.
- Shapiro Ben David S, Potasman I, Rahamim-CohenRate of recurrent guillain-barré syndrome after mrna covid-19 vaccine bnt162b2. JAMA Neurol. 2021;78(11):1409.
- Huang Z, Su Y, Zhang T, Xia A review of the safety and efficacy of current COVID-19 vaccines. Front Med. Published online February 3, 2022.
- World Health Organization. WHO Coronavirus ( COVID-19 ) The World Health Organization. Available from: https://covid19.who.int. Accessed 25 Dec 2021.
|